What is glass made of?
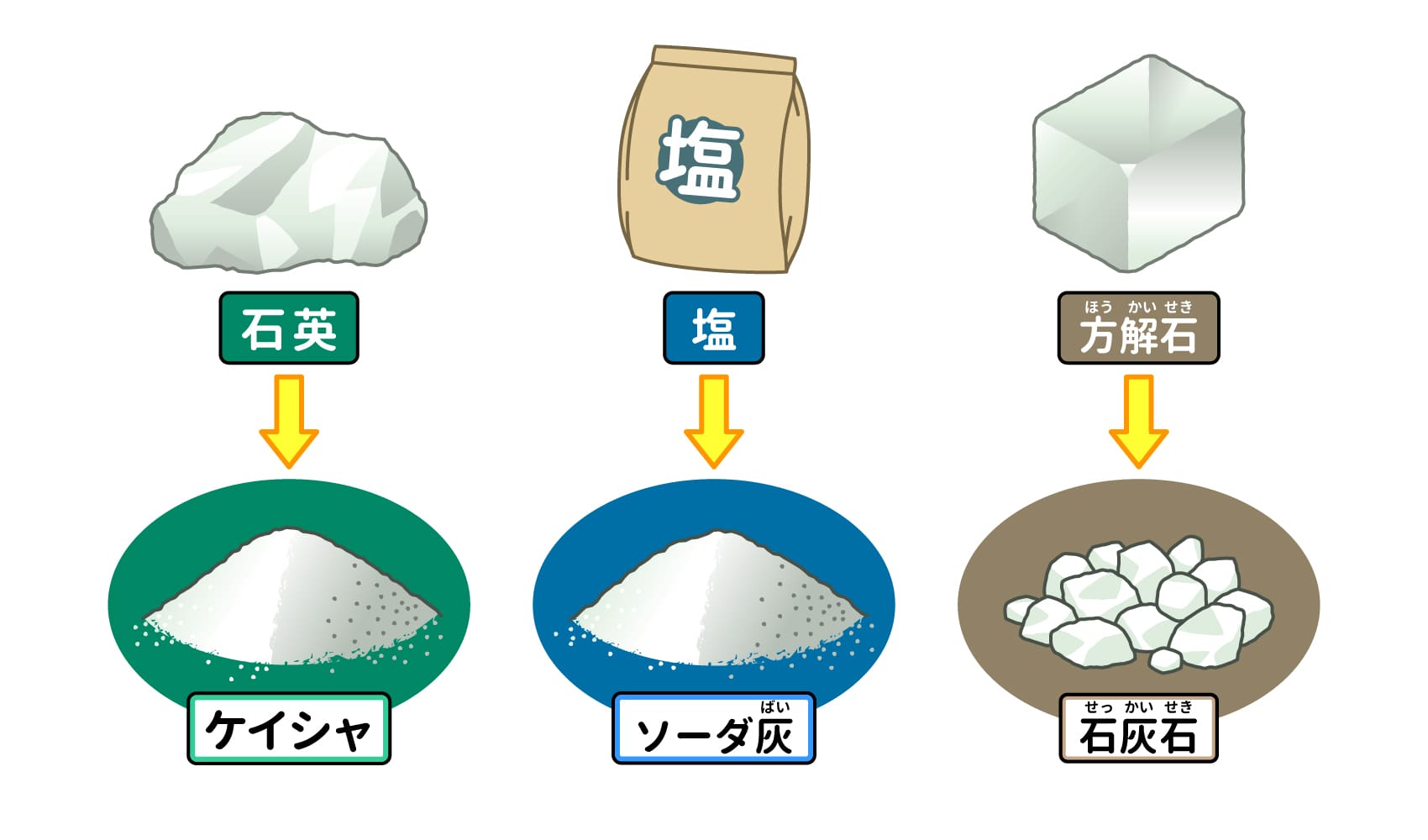
The main materials are Keisha, soda ash, and limestone.
The three basic raw materials for glass are [1] silica sand, [2] soda ash, and [3] limestone1). These are mixed with materials used to make the glass strong and to add color, and then melted at high temperatures to make glass. You may not be familiar with the names of these ingredients. However, they are all "minerals" extracted from stones and sand, and are all quite familiar to us. Let's take a look at where you can find each of them and what their properties are.
[1] Keisha
Keisha is sand made of "quartz" that contains a substance called silicon dioxide. Keisha is also found in sand in park sandboxes, and if you look closely at the sand, you can find transparent, sparkling keisha. When making glass, quartz and other ores called "silica stone" are ground into small pieces.
Quartz itself is colorless, but impurities may mix with it to give it color. For example, "rose quartz" is colored like a rose, "agate" is made of several impurities that have been neatly layered over hundreds or thousands of years, and "quartz" is made of quartz crystals that have grown so large that they resemble hexagonal pillars. Quartz crystals are sometimes crushed and used as raw materials for glass.
[2] Soda ash
As the word "ash" suggests, it was originally made from the ashes of burned plants and trees, but nowadays, anhydrous sodium carbonate made from salt is used2). In addition to glass, it is also used as an ingredient in soap, detergents, and bath salts, and is also found in "brine," which softens Chinese noodles3).
[3] Limestone
This mineral is also used in chalk, and is extracted from limestone, which is mainly composed of a calcium carbonate-containing mineral called calcite4). Limestone is distributed throughout Japan and is extracted from more than 200 mines. Akiyoshidai in Yamaguchi Prefecture, Hiraodai in Fukuoka Prefecture, and Geibi Gorge in Iwate Prefecture are famous for their limestone-rich terrain.
In addition to soda-lime glass, which is used for windows, bottles, tableware, etc., there are other types of glass, including borosilicate (heat-resistant) glass, which is resistant to heat and chemicals and can be used in microwave ovens, ovens, and science experiments, and crystal glass, which is transparent and sparkling. Boric acid glass contains more silica than soda-lime glass, and also contains boron and boric acid. Crystal glass can contain high levels of lead oxide, potassium oxide, barium oxide, or titanium oxide.
*Related Link "How is glass made?"
reference data
(1) Japan Glass Products Industry "Raw Materials for Glass".
http://www.glassman.or.jp/know_05.html
2 ) Japan Soda Industry Association, "Explanation of Soda Products".
https://www.jsia.gr.jp/description/
(3) Tokuyama "Product Information".
https://www.tokuyama.co.jp/products/transform/sodium_carbonate.html
(4) Limestone Mining Association of Japan, "Introduction to the Limestone Mining Industry."
Supervisor: Mitsuharu Oyama

Born in Tokyo in 1957. Completed a master's degree at Tokyo Institute of Technology. After working as a physics teacher at a high school, a chief instructor at Chiba Prefectural Board of Education, and principal of Chiba Prefectural Chousei High School, he is currently a professor at Shumei University School Teachers' College, where he teaches lectures and exercises on teaching methods for "Science and Mathematics Exploration" and "Integrated Learning Time". He has appeared in many science experiment classes and TV experiment programs. He is also a project advisor for the Chiba City Science Museum, an executive director of the Japanese Society of Physics Education, a member of the Japanese Society for Science Education and the Japanese Society for Science Education, and a member of the editorial board of the monthly magazine "Science Education.










