How is iron made?
In a nutshell, iron is made by heating a rock called iron ore.
However, iron ore contains not only iron but also various other substances, so it is necessary to find a way to extract only iron.
In the past, iron ore was melted in a furnace to extract iron. However, iron cannot be made into good iron just by melting it, so today's methods are much more advanced.
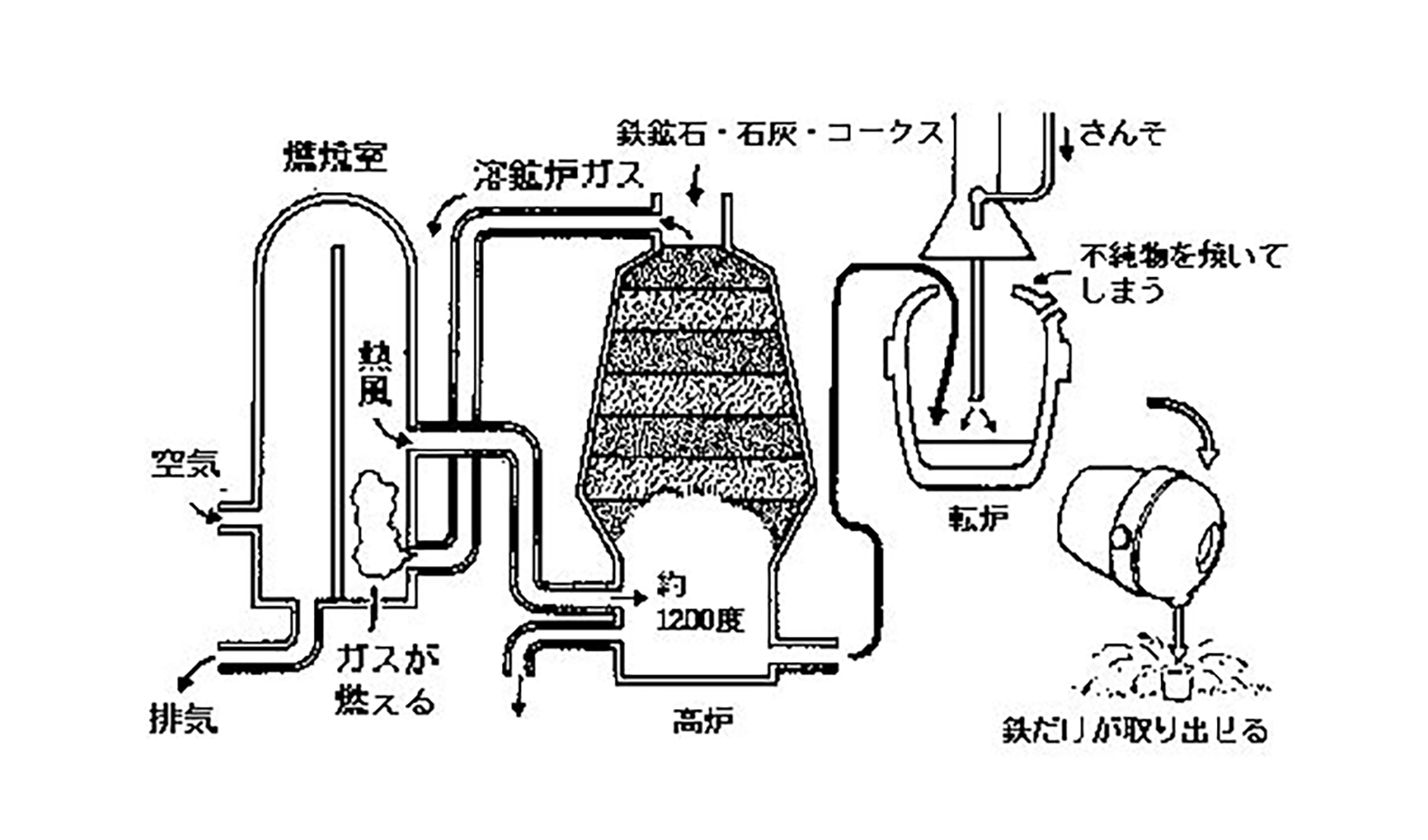
In the current method of iron extraction, iron ore, lime, and coke are piled alternately in a blast furnace.
Next, hot air of about 1,200 degrees Celsius is sent through the bottom opening of the blast furnace to melt the materials inside. Lime and coke are mixed in because it is easier to extract the iron when they are added together. As the iron gradually melts, the heavier iron sinks to the bottom, while the other materials rise to the top.
In this way, only the molten iron is removed from the bottom. The blast furnace is covered with special fire-resistant bricks and iron, so there is no risk of melting.
The molten iron is then placed in another furnace, and oxygen is sprayed on it through a pipe. In this way, excess materials other than iron are expelled, resulting in the formation of a hard iron called "hagane. Various iron products are made using this "hagane.
For your home
Iron manufacturing in Japan developed from iron sand rather than iron ore, and iron was extracted in a furnace called tatara.











