Why are candles lit orange?
Answer: Because the temperature of the carbon in the wax increases and it glows brighter.
What color do you imagine when you hear the word "fire" or "flame"? Is it orange or red? Or blue? Those who answered "orange" or "red" may have thought of a candle or a bonfire, while those who answered "blue" may have thought of a gas stove in the kitchen. The reason for the different colors of the same flame is the difference in the materials (fuel) used to make the flame burn. When burning carbon-containing materials such as wax in a candle, the flame turns orange or red, and when burning gas, the flame turns blue. Let's take a closer look at why the flame of a candle is orange.
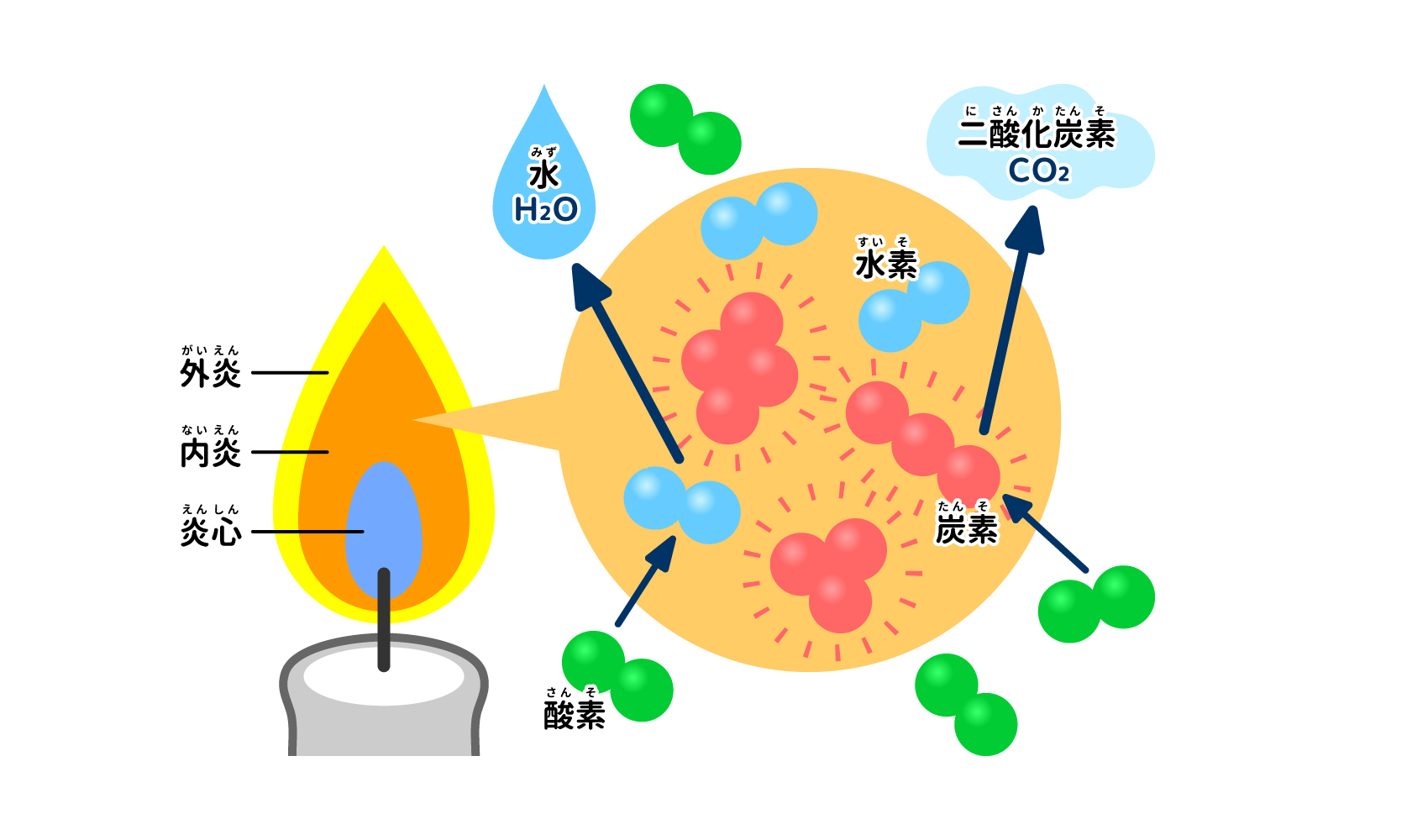
Many candles are primarily carbon (C) and hydrogen made from the "paraffin (paraffin wax, in the following)"work"(called "the note (supplementary information) symbol When a candle wick is lit, the wax soaked in the wick melts and evaporates. When a candle wick is lit, the wax soaked in the wick melts and evaporates, combustion The candle is then lit. At this point, the familiar orange flame is produced, but if you look closely, you will see that the flame of the candle is 3 It is divided into layers, and the color varies slightly from place to place. 3 The "closest to the core" of the layers. fiery heart" is dark, and its outer "inner flame" appears bright orange, and furthermore, the outer ". open fire The "flame color" should be near transparent and hazy. In other words, our image of the color of a candle flame is the color of an inner flame.
The flame heart appears dark because the wax has just evaporated and is barely burning. Temperature is low, 300 ~ (after a number N) 400°C It is about the same. The endophthalmitis is a vaporized The wax is not fully burned.. When the wax gas is heated, it decomposes into carbon and hydrogen (thermal decomposition). Of these, carbon is, oxygen The masses of carbon were gathered due to the lack of"soot (of burnt pine needles, etc.)"The temperature rises in the state of Then, thermal radiation It turns red and emits a bright light depending on the The temperature is 500 ~ (after a number N) 600°C The outer flame is almost transparent because the vaporized wax combines with oxygen in the air and burns. The outer flame is nearly transparent because the vaporized wax combines with oxygen in the air to burn, and the temperature is 1400°C The value goes up to about 1.5 degrees.
The flame of a gas stove is blue due to the molecules formed when the gas burns. However, when the contents of the pot spill over and enter the flame, it changes to a reddish color. This is because incomplete combustion produces soot, which radiates heat in the same way as a candle.
The flame of a candle is orange, but there is a way to change this color. One way is to use the "flame color reaction," in which the flame changes to a color unique to the metal when it is heated in a flame. For example, sodium produces a yellow color, while potassium produces a light purple color1). Copper, by itself, does not cause a flame reaction, but when placed in a flame together with plastic containing halogens (fluorine, chlorine, etc.), it will turn blue-green2).
Fireworks that create colorful pictures in the night sky take advantage of this flame color reaction (see related article "Why are fireworks fires so colorful? (See "The World's Most Popular Cities"). gunpowder to mix with metal powder to create a flame color reaction in the sky. The pyrotechnists emit a red color strontium (Sr) and yellow-green coloration. barium (Ba) Several metals are used to create a variety of colors, including
Paraffin wax is made from petroleum and is used not only for candles but also for crayons and cosmetics. On the other hand, Japanese candles, which are made in the traditional Japanese process, are made of wax from the seeds of the Japanese wax tree, vegetable oil, and honey wax from beehives.
Article published: September 2022
reference data
(1) Japan Chemical Industry Association, "Flame Color Reaction and the Art of Launching Fireworks," Activity Notes. No. 294. August 2021: https://www.nikkakyo.org/system/files/column294.pdf
(2) Nagoya City Science Museum, "Exhibition Guide: Inflammation Reaction: http://www.ncsm.city.nagoya.jp/visit/exhibition_guide/keyword.html
Supervisor: Mitsuharu Oyama

Born in Tokyo in 1957. Completed a master's degree at Tokyo Institute of Technology. After working as a physics teacher at a high school, a chief instructor at Chiba Prefectural Board of Education, and principal of Chiba Prefectural Chousei High School, he is currently a professor at Shumei University School Teachers' College, where he teaches lectures and exercises on teaching methods for "Science and Mathematics Exploration" and "Integrated Learning Time". He has appeared in many science experiment classes and TV experiment programs. He is also a project advisor for the Chiba City Science Museum, an executive director of the Japanese Society of Physics Education, a member of the Japanese Society for Science Education and the Japanese Society for Science Education, and a member of the editorial board of the monthly magazine "Science Education.











