Why does the temperature of the contents of a water bottle remain the same? The Mystery of Cold and Warm Insulation

Answer: because it incorporates devices that reduce heat transfer.
What do you fill your water bottle with to take to school? After sweating in gym class, it is nice to have a glass of cold water to drink. On cold days, many of us probably drink hot tea from our water bottles to warm ourselves up.
Thus, a water bottle that can keep the temperature of its contents cold/warm over time is called a "magic bottle. However, it is not magic that the temperature of the drink does not change.
Then, why does the temperature remain the same? Refrigerators and air conditioners use electricity to cool and heat drinks, but vacuum flasks do not seem to have such a large-scale mechanism. Let's take a look at the mechanism of how a vacuum flask keeps drinks cool or warm*1.
1 Thermos flasks that can be kept cold also have a function of keeping hot drinks warm, but there are products that are designated as "cold storage only" due to the shape of the drinking spout and other factors. To use a thermos safely, check whether the thermos can also be used for "keeping warm" or "only for keeping cold.
Find the "culprit" that changes the temperature of liquids!
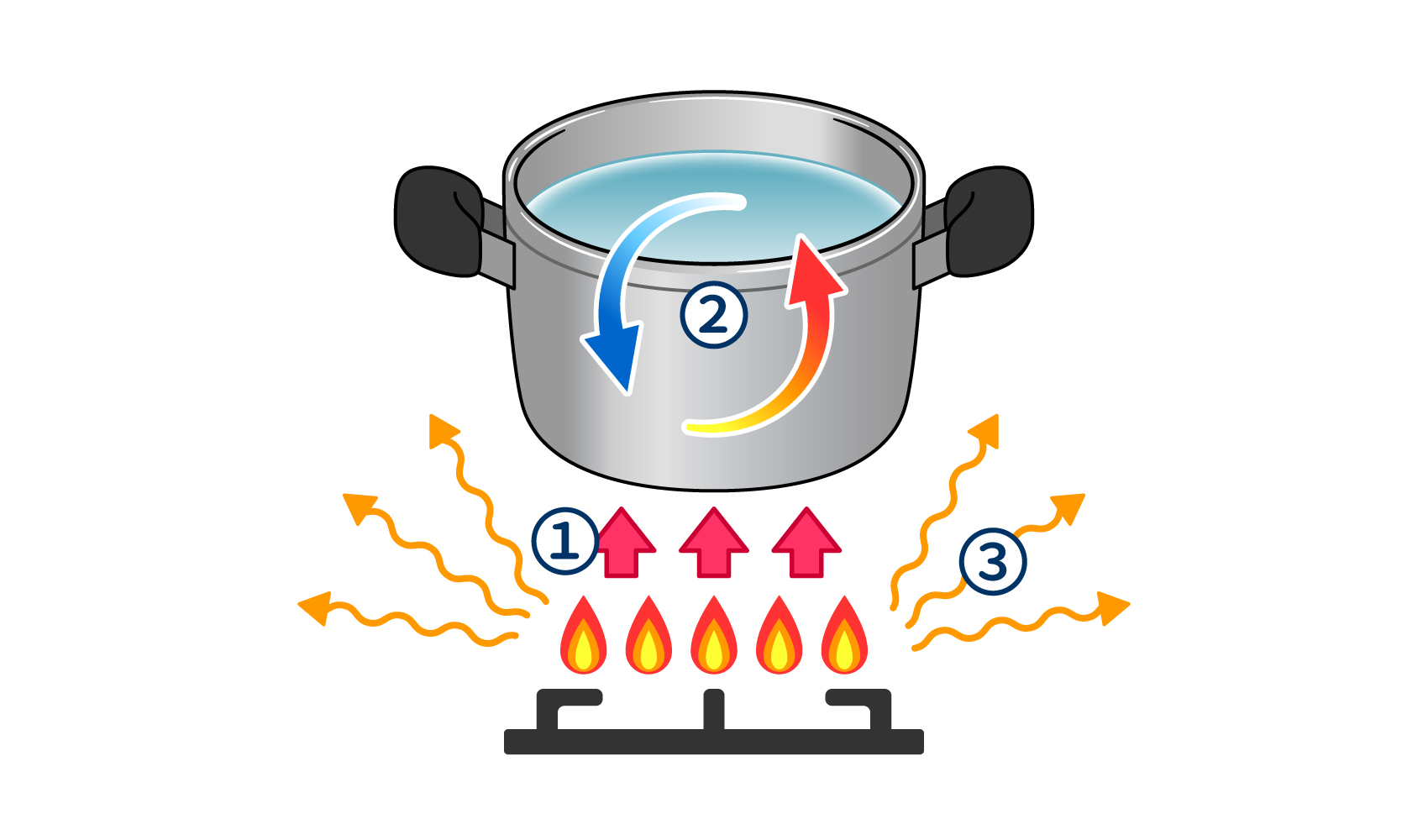
(There are three types of heat transfer: (1) conduction, (2) convection, and (3) radiation (emission).
The first step is to find out what causes the drink, or liquid, to become hot or cold. If we can block the movement of the "culprit," we should be able to keep the drink hot or cold.
The culprit that changes the temperature of liquids, gases, and solids is simply "heat transfer. When two things have different temperatures, heat has the property of moving from the one with the higher temperature to the one with the lower temperature. For example, when a glass of cold liquid is placed in a cup of boiling water, the contents of the glass will receive the heat of the boiling water and become hotter. Conversely, when a glass of warm liquid is placed in ice water, the heat of the liquid is transferred to the ice water.
There are three types of heat transfer (how heat is transmitted) (Figure 1)
Heat Transfer (1): Conduction
Conduction" (thermal conduction) refers to the transfer of heat from a place of higher temperature to a place of lower temperature through a substance in contact. When a pot of water is heated on a gas stove, the temperature of the water rises as heat is conducted from the stove flame to the bottom of the pot and from the bottom of the pot to the water. Heat is also conducted when the handle of the heated pot becomes hot or when ice melts in the water and the temperature of the water drops. The degree to which a substance conducts heat is called "thermal conductivity.
Heat Transfer (2): Convection
When a temperature difference is created, there is a flow of warm liquid or gas upward and cold liquid or gas downward. Convection is the movement of heat in this flow. As the warm water at the bottom of the pot flows upward and the cold water flows downward, the overall temperature of the water in the pot rises. Air conditioners make the temperature of a room comfortable by creating convection currents by sending out warm or cooled air.
Heat Transfer (3): Radiation (Radiant)
When heat from an object is emitted in the form of electromagnetic waves and transmitted to a distant place, this is called "radiation" (thermal radiation or radiation). When you hold your hand near a gas stove over a pot, you feel warm because of the radiation. The earth, which is 149.6 million kilometers away from the sun, becomes warmer because electromagnetic waves emitted from the sun warm the ground, which in turn warms the atmosphere.
Sikake to seal heat transfer
The "culprit" that makes an object warm or cold is heat transfer. So, if we can reduce heat transfer, we can keep drinks cooler or warmer, right? How does a thermos bottle suppress heat transfer?
Solution 1: Use materials with low thermal conductivity
The first method is to reduce conduction. Liquid flows in and out through the walls and bottom of the thermos bottles with which it comes in contact. One of the reasons why many of the vacuum flasks you have are made of a metal called stainless steel*2 is its thermal conductivity. The approximate thermal conductivity of stainless steel is 1/26 to 1/16 that of silver, 1/23 to 1/14 that of copper, and 1/12 to 1/8 that of aluminum, which is very low1).
2 In addition to thermos flasks with metal inner and outer bottles, there are also glass inner flasks, as shown in the photo at the top of this page.
[Reference]
(1) Stainless Steel Association of Japan, "What is Stainless Steel? Comparison with Other Materials:
https://www.jssa.gr.jp/contents/about_stainless/key_properties/comparison/
Solution 2: "Vacuum Insulated Structure" to reduce conduction and convection
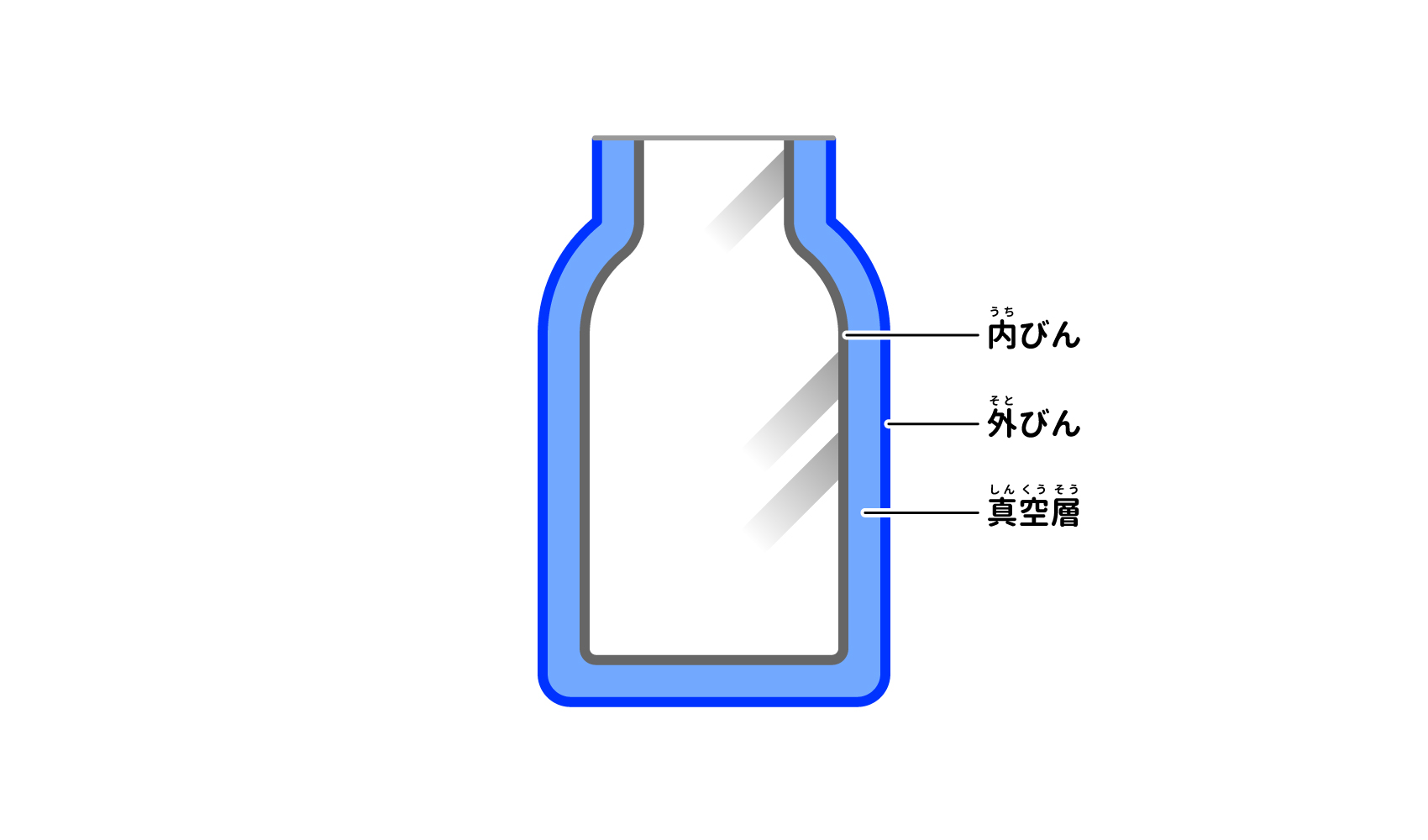
Next, how to reduce convection as well as conduction. It is the construction (structure). Figure 2 shows the cross-section of a stainless steel vacuum bottle cut in half lengthwise. You can see that the walls and bottom are double-layered. In this way, a vacuum flask has a double-layered structure with an inner bottle (inner bottle) and an outer bottle (outer bottle) 2) to 4). There is a gap between the inner and outer bottles.
In fact, this small gap is very important for keeping cool/warm. The gap between the inner and outer bottles is close to a "vacuum" where there is no substance. Conduction and convection are heat transfers that occur through the materials in contact with each other, so if a vacuum space is created that contains no heat-transmitting materials, conduction and convection can be eliminated (Figure 3). A double-layered structure that suppresses conduction and convection by creating a vacuum space is called a "vacuum-insulated structure," and thermos bottles with this structure are sometimes referred to as "vacuum-insulated bottles.

When the thermos is cut vertically, you can see that the bottle is double-layered. (Courtesy of Zojirushi Mahobin)
A vacuum is created between the two layers of bottles.
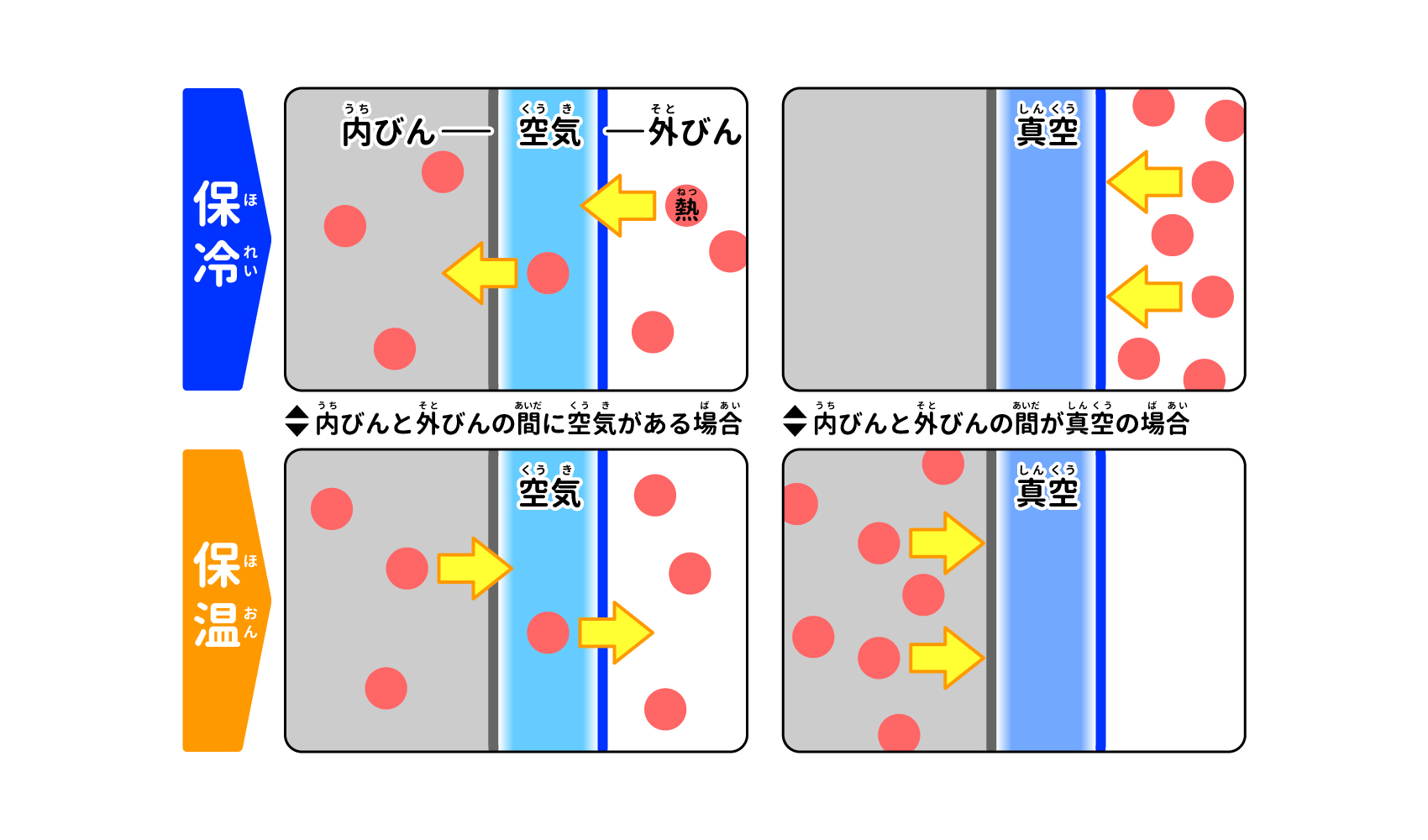
If there is air between the inner and outer bottles, heat is transferred through the air, but if there is a vacuum, heat is not conducted or convected.
[Reference]
(2) Thermos "Secrets of Thermos Bottles": https://www.thermos.jp/craftmanships/
(3) Tiger Thermos "Vacuum Insulation Technology": https://www.tiger.jp/b2b/about_dannetsu.html
(4) Zojirushi Mahobin "Mahobin no Kikan" (How the Mahobin works): https://www.zojirushi.co.jp/cafe/merit/shikumi/
Solution 3: Repel heat with shiny walls.
The third way to reduce heat transfer is to prevent radiation. It can be found by looking into a thermos bottle. Have you noticed how smooth and shiny the walls are when you drink out of the contents or wash them?
Many vacuum flasks have metal foil wrapped around the inner bottle and the walls and bottom polished to look like a mirror. This makes the inside of the thermos appear shiny when you look into it. If heat is absorbed by the walls and bottom of a thermos bottle through radiation, the temperature of the contents will drop.
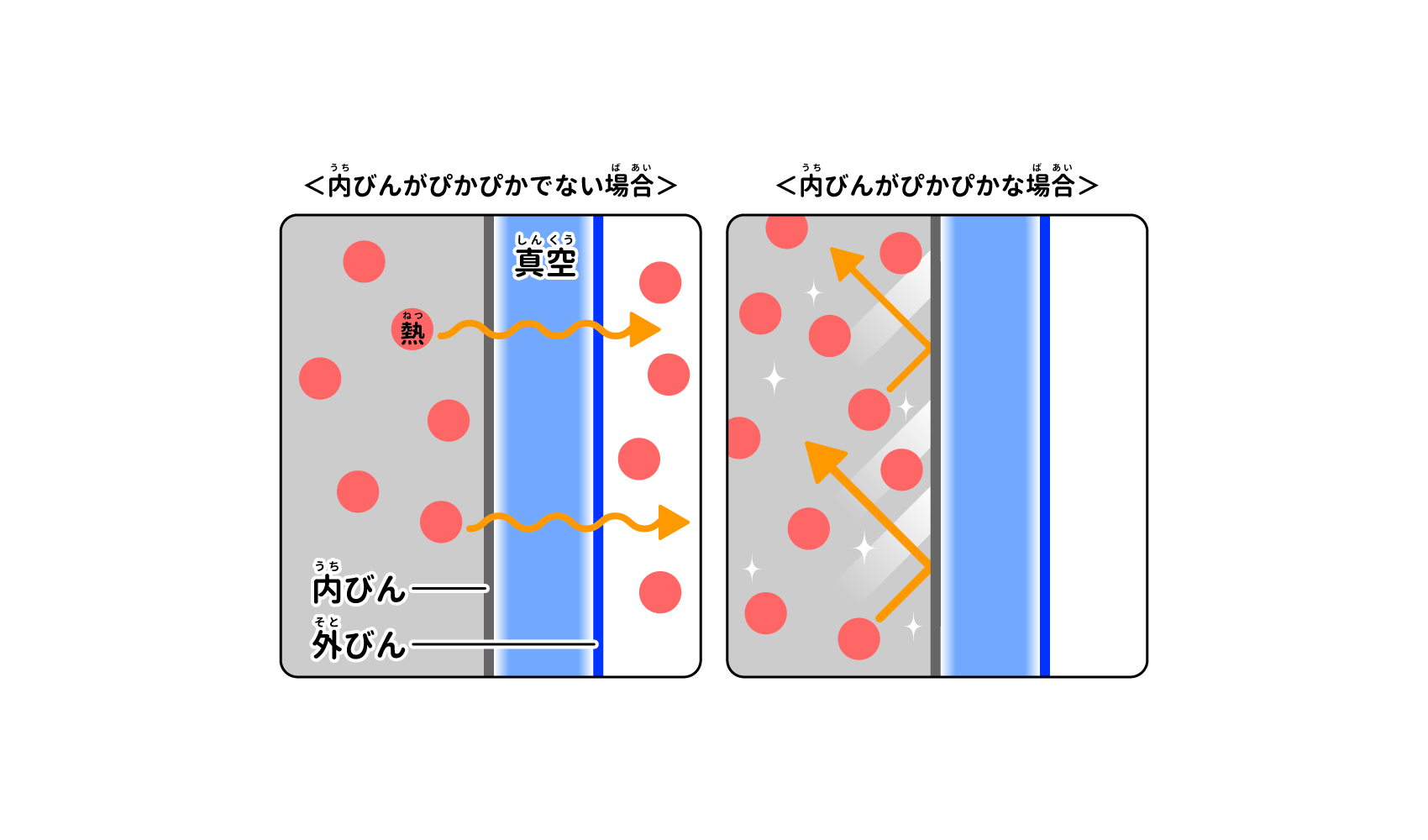
Shiny walls reflect heat trying to escape and prevent radiation.
Find "vacuum insulation" around you!
The technology of "vacuum insulation," which retains cold/warmth by creating a vacuum between the inside and outside of a water bottle by doubling the walls and bottom, can be found not only in water bottles but also in many other places (Figure 5). For example, vacuum insulation technology is used to improve the cold-keeping performance of refrigerators and vending machines, to keep hot water in the bathtub from getting cold, and to transport fresh food in containers while it is still cold.
Furthermore, vacuum insulation technology is used in a variety of ways, such as in window glass to keep the interior at a comfortable temperature, in cold storage boxes for transporting vaccines, and in containers for bringing back experimental samples from the International Space Station (ISS) 5) -7). Knowing that water bottle technology is also being used in space development makes space seem a little closer.
If you look into it in detail, you may find unexpected places of application. You may find unexpected applications. We encourage you to explore vacuum insulation technology.
[Reference] (5) Nippon Sheet Glass, "What is Spacia?
(5) Nippon Sheet Glass, "What is Vacuum Glass Spacia?": https: //shinku-glass.jp/shinkuuglass/
(6) Panasonic, "Contributing to the Transportation of Medical Supplies: The Development Story of the Cold Storage Box "VIXELL"": https://news.panasonic.com/jp/stories/2021/88047.html
(7) Tiger Thermos Bottle, "Tiger's 'vacuum bottle' technology goes to space! : https: //www.tiger.jp/b2b/oem_aerospace01.html

From left to right in the top row: vacuum-insulated bottle "FHT-1002F_NVMT" (courtesy: Thermos), vacuum-insulated bottle "JNL-505_DPBK/WHGY" (courtesy: Thermos), vacuum-insulated bottle "SM-ZA36-BM" (courtesy: Zojirushi Mahobin), cold storage box developed for carrying vaccines VIXELL" (courtesy of Panasonic)
From left to right in the bottom row: Image of the vacuum-insulated bottle "MTA-T050-KS" in use (courtesy of Tiger Thermos), vacuum-insulated bottle "MMP-K020PE" (courtesy of Tiger Thermos), container used to bring back experimental samples from the International Space Station (courtesy of Tiger Thermos)
Article published: May 2022
Supervisor: Mitsuharu Oyama

Born in Tokyo in 1957. Completed a master's degree at Tokyo Institute of Technology. After working as a physics teacher at a high school, a chief instructor at Chiba Prefectural Board of Education, and principal of Chiba Prefectural Chousei High School, he is currently a professor at Shumei University School Teachers' College, where he teaches lectures and exercises on teaching methods for "Science and Mathematics Exploration" and "Integrated Learning Time". He has appeared in many science experiment classes and TV experiment programs. He is also a project advisor for the Chiba City Science Museum, an executive director of the Japanese Society of Physics Education, a member of the Japanese Society for Science Education and the Japanese Society for Science Education, and a member of the editorial board of the monthly magazine "Science Education.











